 
If the FHR result is distrustful, please use other methods such as stethoscope to verify immediately. WARNING : This device is not intended for treatment. Responsibility of the Manufacturer EDAN only considers itself responsible for any effect on safety, reliability and performance of the equipment if: Assembly operations, repairs are carried out by persons authorized by EDAN, and the device is used in accordance with the instructions for use. The information contained in this document is subject to change without notice. No part of this document may be photocopied, reproduced or translated to another language without prior written consent of EDAN. 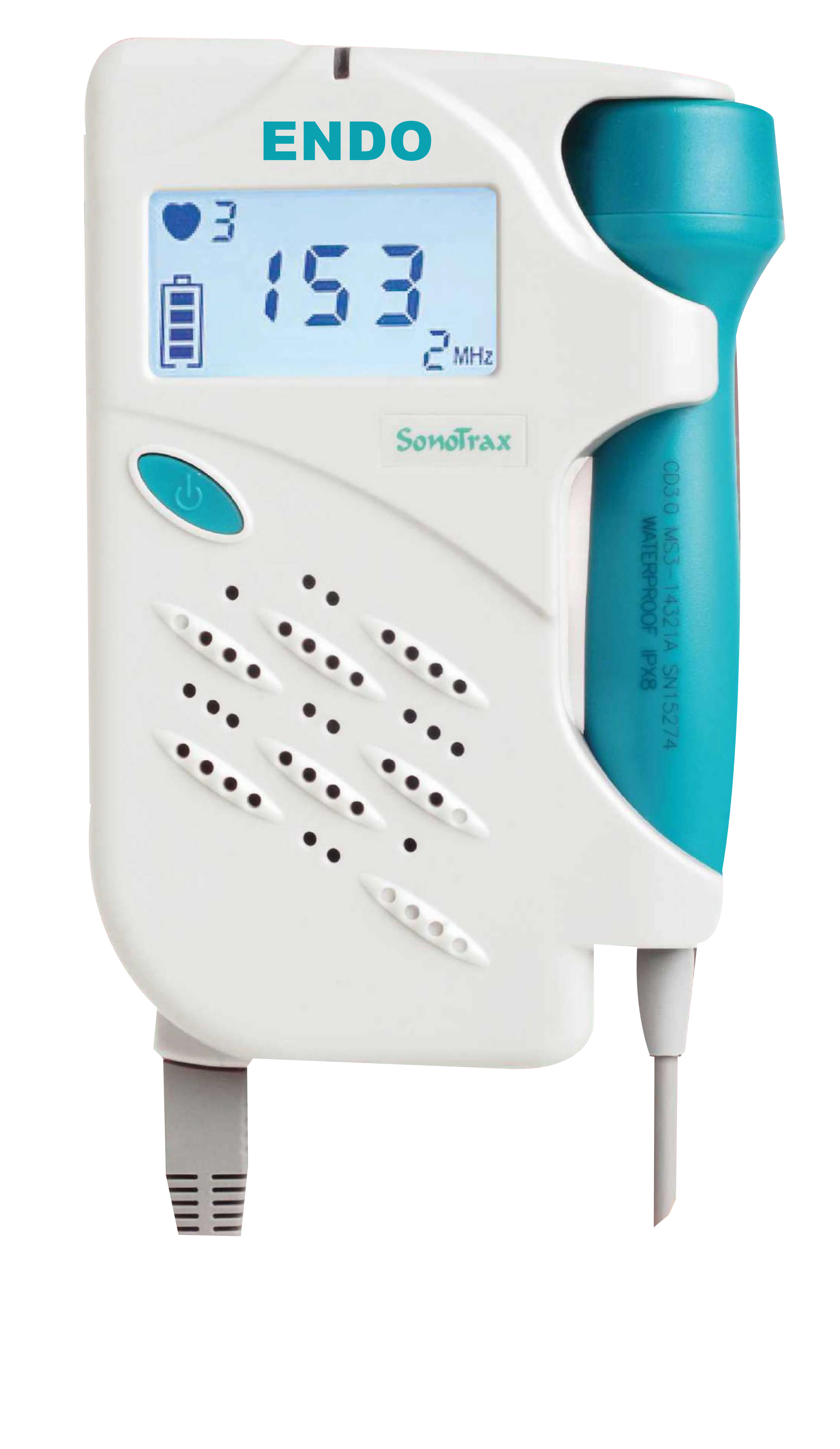 
EDAN assumes no responsibility for any error that may appear in this document, or for incidental or consequential damage in connection with the furnishing, performance or use of this material. (hereinafter called EDAN) makes no warranty of any kind with regard to this material, including, but not limited to the implied warranties of merchantability and fitness for a particular purpose. All rights reserved.Īttention Edan Instruments, Inc. Notified Body: TÜV SÜD Product Service GmbH, Ridlerstr 65, D-80339 MÜnchen, Germany.Ĭopyright © Copyright Edan Instruments, Inc. All supporting documentation is retained at the premises of the manufacturer.ĭIRECTIVES General Applicable Directives: Medical Device Directive: COUNCIL DIRECTIVE 93/42/EEC of 14 June 1993 concerning medical devices (MDD 93/42/EEC). SONOTRAX II, SONOTRAX II Pro Classification (MDD, Annex IX): IIa We herewith declare that the above mentioned product(s) meet the transposition into national law, the provisions of Council Directive 93/42/EEC of 14 June 1993 concerning medical devices - as amended by Directive 98/79/EC on in vitro diagnostic medical devices. GmbH (Europe) Addr: Eiffestrasse 80 D-20537 Hamburg Germany Product: Ultrasonic Pocket Doppler Model: SONOTRAX Lite, SONOTRAX Basic, SONOTRAX Basic A, SONOTRAX Pro, CHINA European Representative:Shanghai International Holding Corp. Addr: 3/F-B, Nanshan Medical Equipments Park, Nanhai Rd 1019#, Shekou, Nanshan Shenzhen, 518067 P.R. EC Declaration of Conformity Manufacturer: EDAN Instruments, Inc. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
